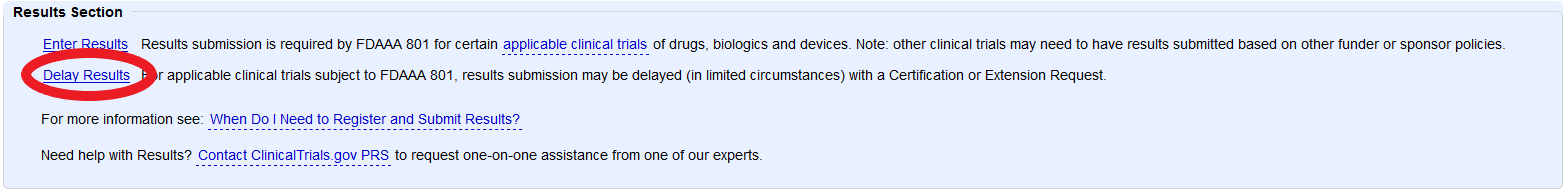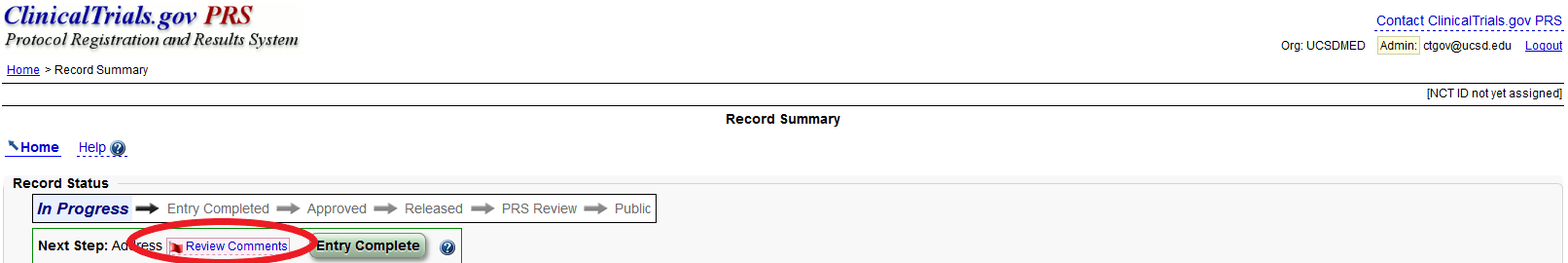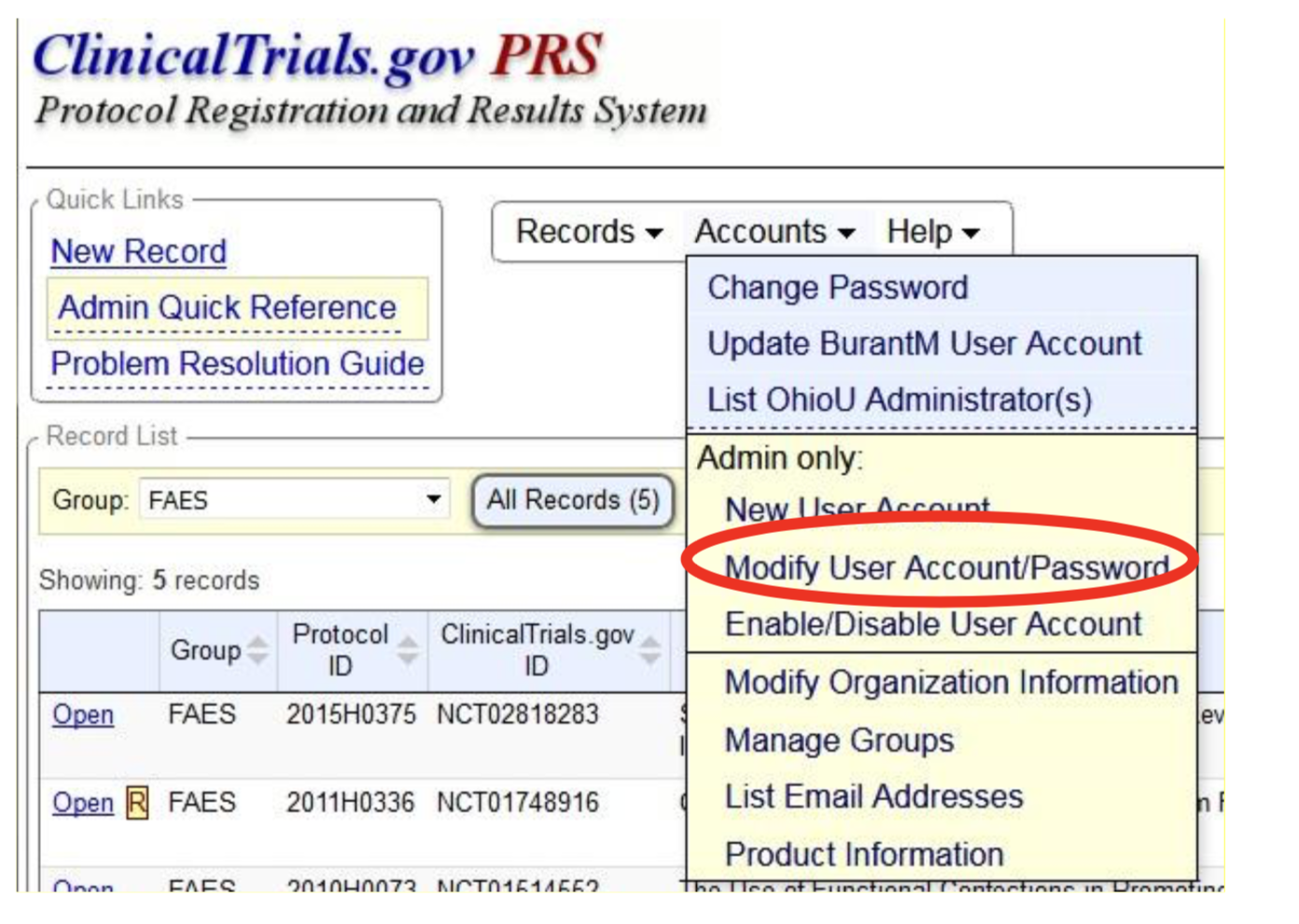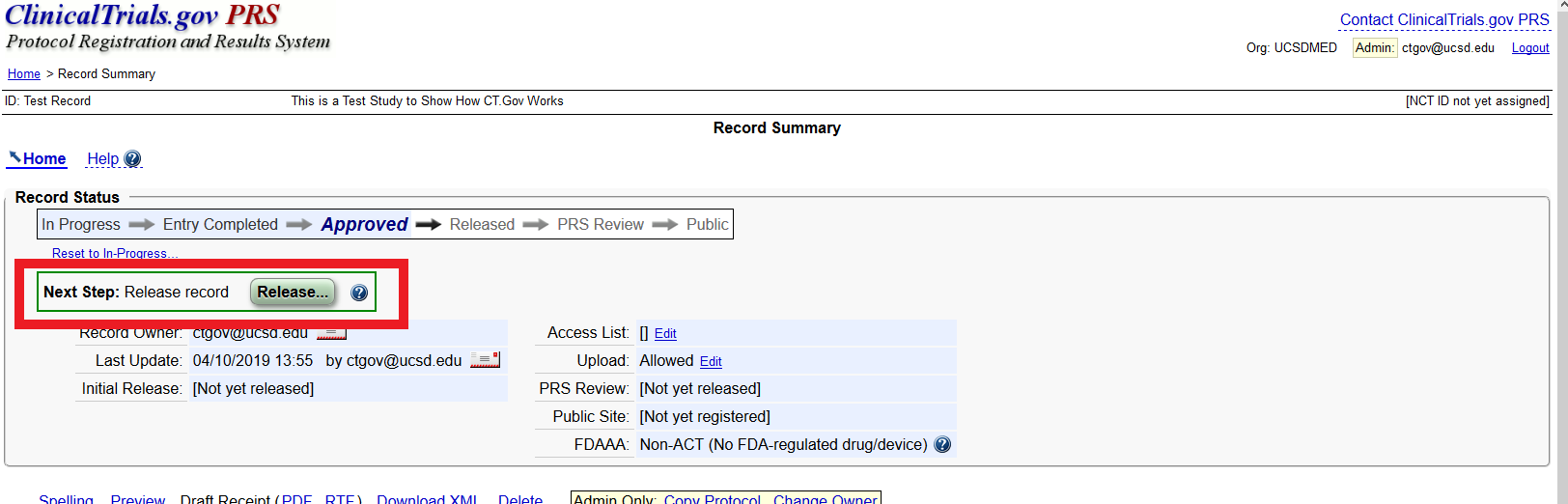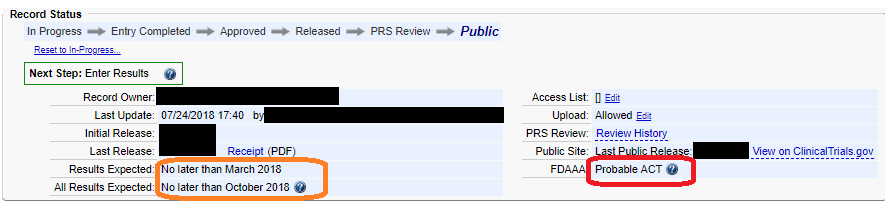
ClinicalTrials.gov Tip of the Week: Seife et al. v. HHS et al.: Results Must be Submitted for “pACTs” to Clinical Trials.gov

ClinicalTrials.gov: How to Register Your Trial - Clinical and Translational Science Institute - University at Buffalo

PDF) ClinicalTrials.gov Brief Title: Cholecalciferol to Improve the Outcomes of COVID-19 Patients ( CARED ) Official Title: Randomized Controlled Trial of High Dose of Vitamin D as Compared With Placebo to Prevent