Book M1: 2022 Mini Pocket-Sized (3" x 5") ICH Guidelines for GCP (E6) – Clinical Research Resources, LLC

Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines by Tom Brody PhD Dr. (2016-03-14): Amazon.com: Books

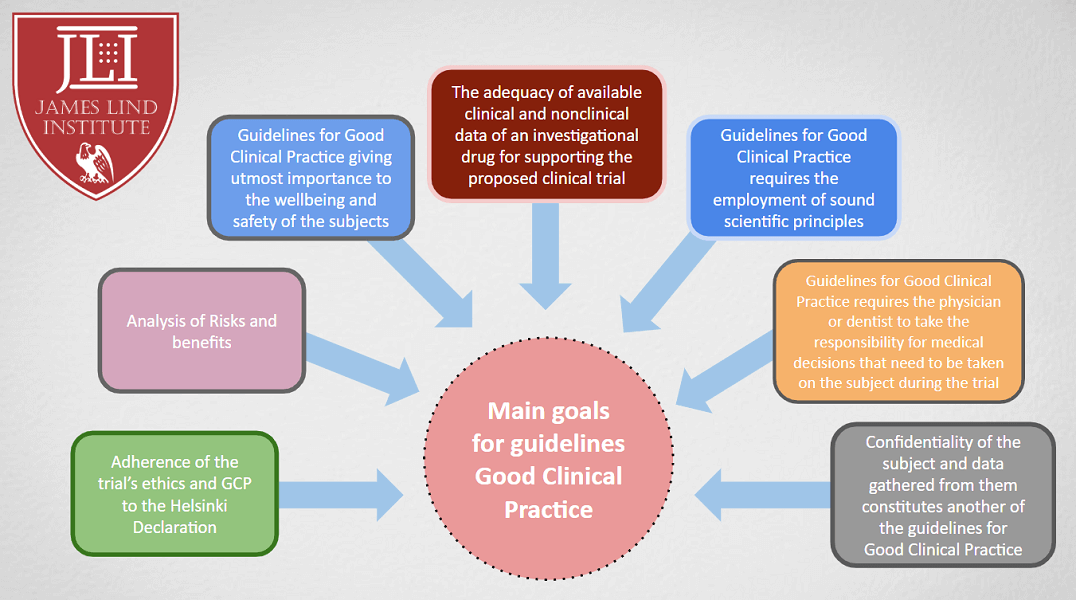
Principles of Good Clinical Practice (GCP) – What is it all about and who is responsible for adherence? GCP and QA All SIAC Call Mar 14, 2008 Munish Mehra, - ppt download

Compliance with the ICH-GCP Guidelines among the Saudi Health Care Professionals: Should Saudi Arabia Conduct Widespread ICH-GCP Training? | Semantic Scholar


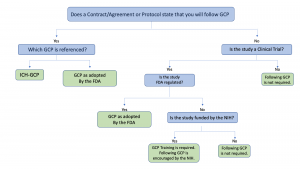



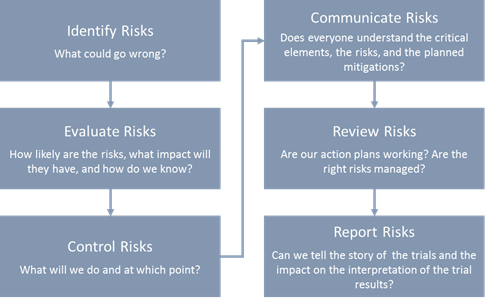






(135).jpg)