Clinical Effectiveness of Idarucizumab in Dabigatran Reversal | Wang | Journal of Cardiology and Therapy

Idarucizumab dosing in patients with excessive dabigatran body burden - British Journal of Anaesthesia
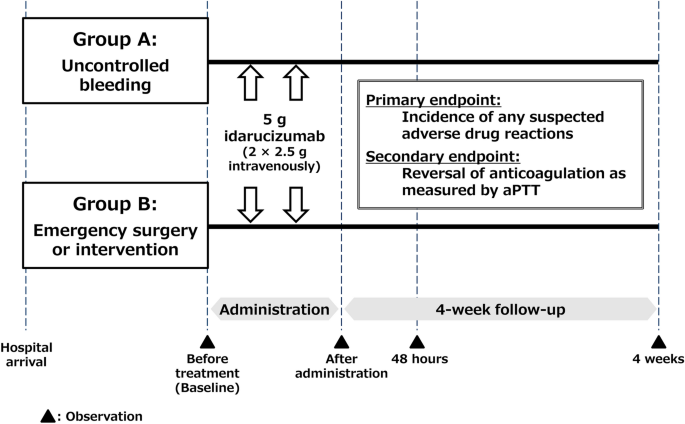
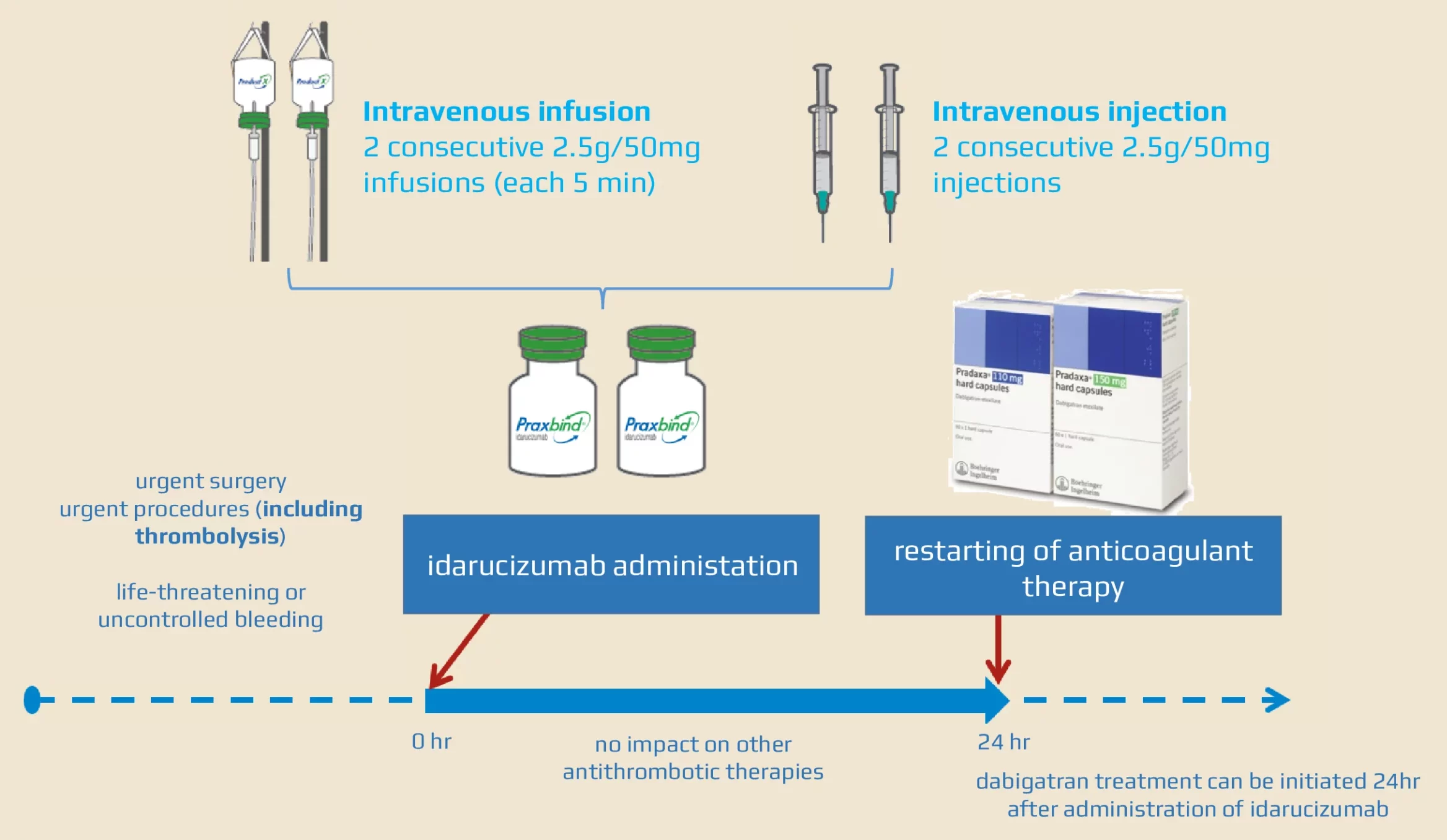
Idarucizumab for Emergency Reversal of Anticoagulant Effects of Dabigatran: Interim Results of a Japanese Post-Marketing Surveillance Study | SpringerLink

Design of the Reversal Effects of Idarucizumab on Active Dabigatran... | Download Scientific Diagram
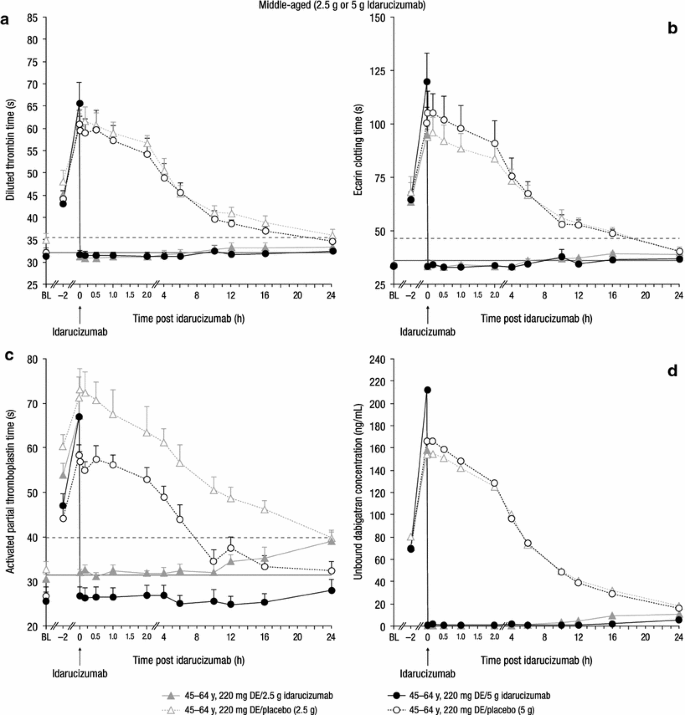
Effect of Age and Renal Function on Idarucizumab Pharmacokinetics and Idarucizumab-Mediated Reversal of Dabigatran Anticoagulant Activity in a Randomized, Double-Blind, Crossover Phase Ib Study | SpringerLink
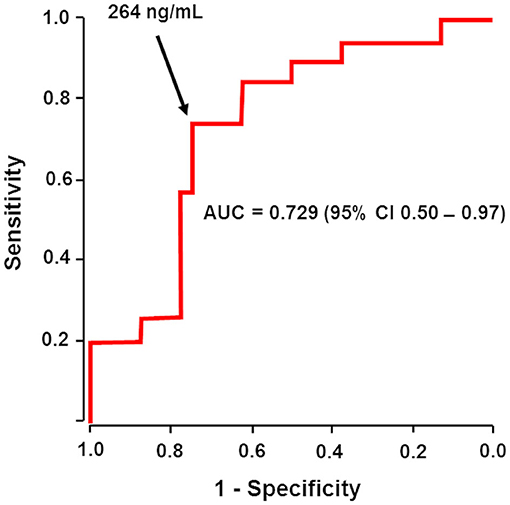
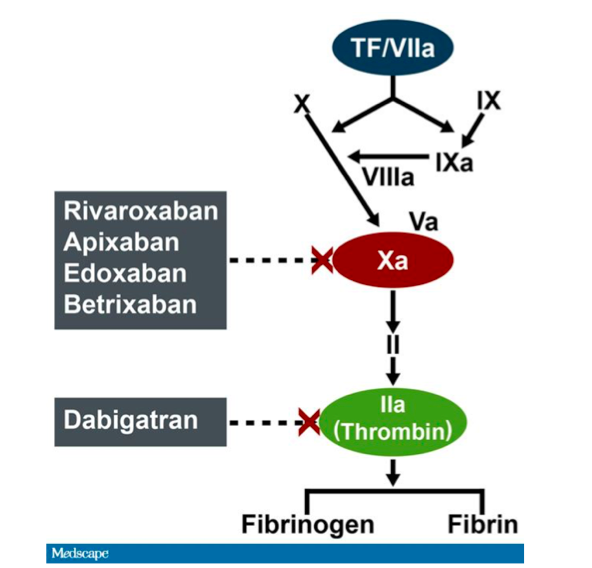
Frontiers | Dabigatran Level Before Reversal Can Predict Hemostatic Effectiveness of Idarucizumab in a Real-World Setting

FDA Approves Praxbind® (idarucizumab), Specific Reversal Agent for Pradaxa® (dabigatran etexilate) | Business Wire

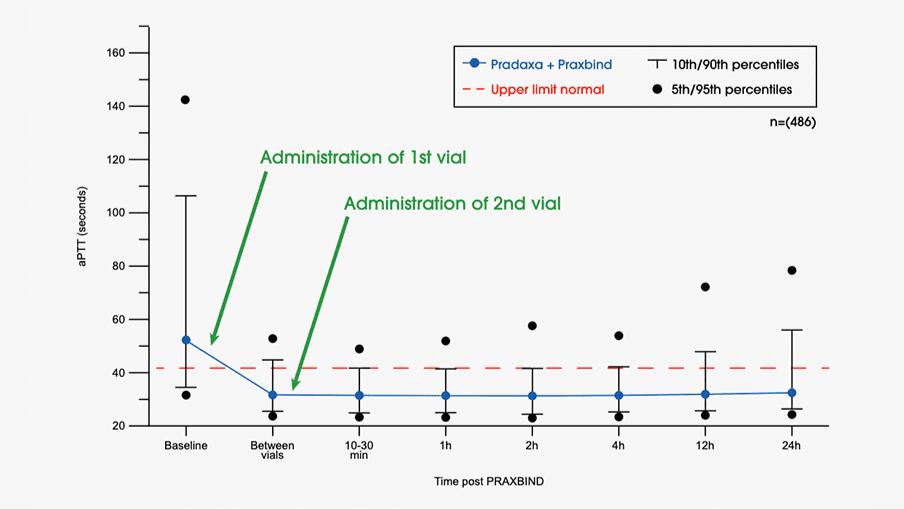
Safety, tolerability, and efficacy of idarucizumab for the reversal of the anticoagulant effect of dabigatran in healthy male volunteers: a randomised, placebo-controlled, double-blind phase 1 trial - The Lancet
Interpretation of idarucizumab clinical trial data based on spontaneous reports of dabigatran adverse effects in the French phar

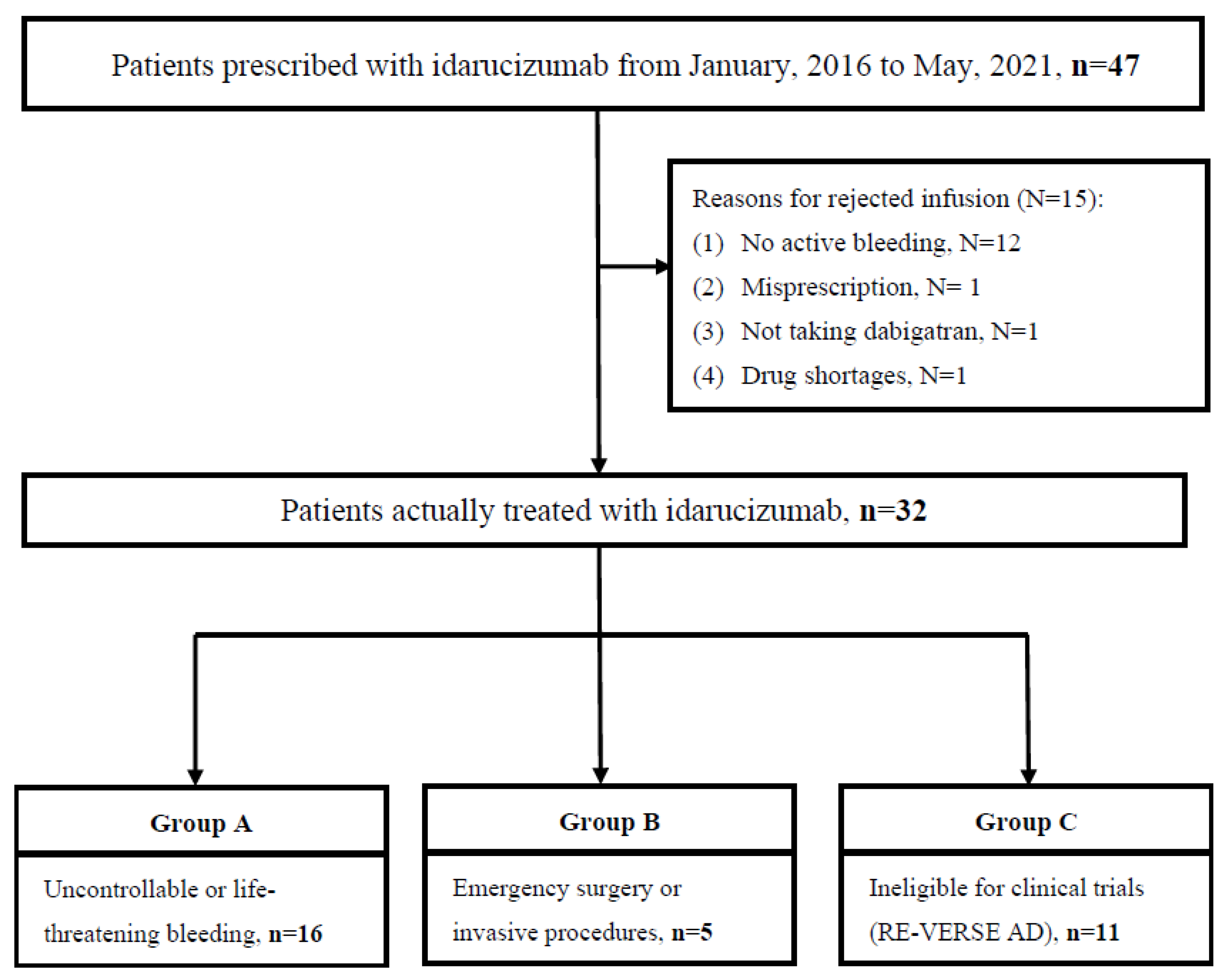
Medicina | Free Full-Text | Effectiveness and Safety of Dabigatran Reversal with Idarucizumab in the Taiwanese Population: A Comparison Based on Eligibility for Inclusion in Clinical Trials

Safety, pharmacokinetics and pharmacodynamics of idarucizumab, a specific dabigatran reversal agent in healthy Japanese volunteers: a randomized study - Research and Practice in Thrombosis and Haemostasis
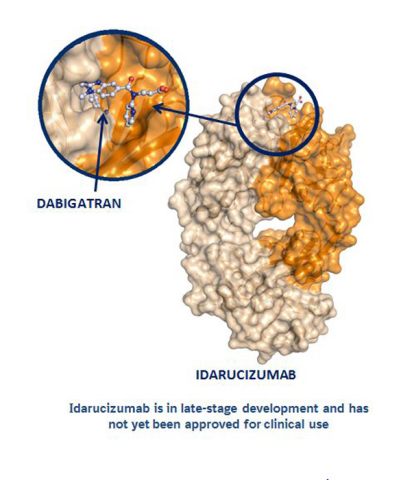
Boehringer Ingelheim submits applications for approval of Idarucizumab, an anti-anticoagulant - Labiotech.eu